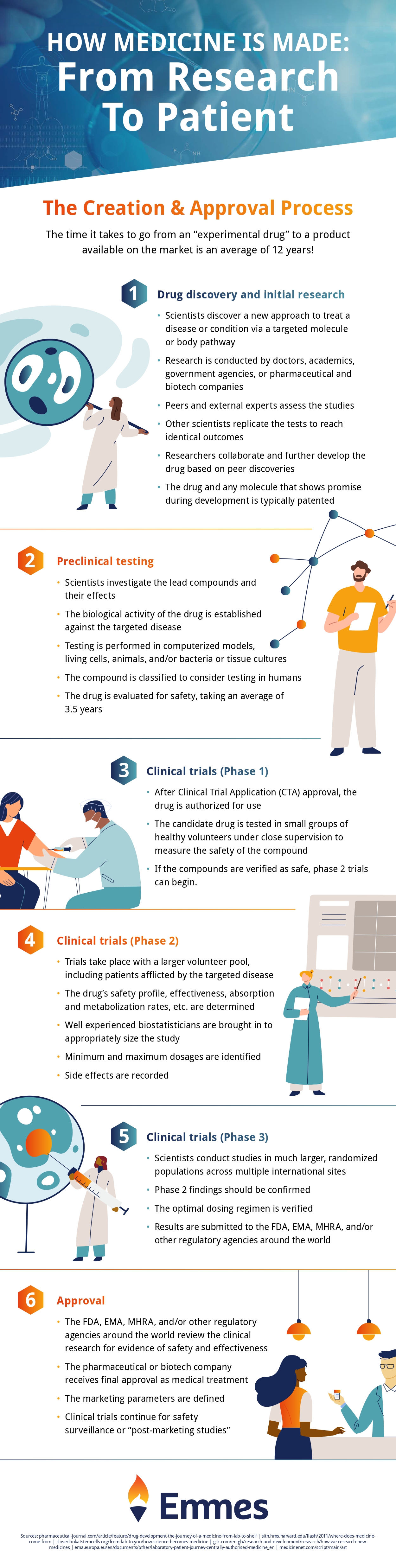
Considerations for Implementing Decentralized Clinical Trials

Business, Technology, Internet and network concept. select on the virtual display: Clinical trial
There has been a huge increase in the adoption of decentralized clinical trials since the pandemic began. The rapid changes in clinical trials will certainly continue beyond the pandemic. The typical centralized clinical trials that cater to investigators at a physical site are slow, expensive, slow, and outdated. On the other hand, Veristat decentralized clinical trials had a completely different clinical trial design. Patients can participate from different physical locations as long as they often report to the investigators.
However, implementing decentralized clinical trial methodologies and technologies is not as easy as it sounds. CROs have to consider some key factors during the clinical trial planning phase to mitigate the complexities. Here are the factors to consider:
Consider a flexible design
A fully decentralized trial might not always be feasible for certain studies and conditions. Therefore, developers must first consider which elements can be decentralized and appropriate for site-based visits. It is also important to consider that DCT could have telemedicine and traditional site visits. With telemedicine, the patients report to the investigators remotely while patients get an in-person review with the traditional visits.
Have a proactive strategy
By adopting a proactive strategy, sponsors can use different investigators and patient networks to reduce the trial start-up time from months to weeks. They can then use centralized remote coordinators and mobile healthcare personnel to maximize the trials and enrich the patient experience. There is increased patient satisfaction and retention with decentralized trials, such as gathering patient input during the development process while using patient-reported outcomes.
Place the needs of the patient first
One major benefit of decentralized clinical trials is that it places the needs of the patients first. However, this approach may be attributed to technology, but tech alone does not equate to patient centricity. Patient-centered trials mean an early and continuous dialogue with the patients, advocates, and caregivers to understand and learn the individual’s patient’s journey, preferences during visits, and trial experiences. Patients also get to voice their challenges early enough during the trial.
The Trial regulators
Although the methods and tools used in decentralized clinical trials are not new, there was a very slow pace to adoption before the pandemic. This could be attributed to an uncertainty around gathering regulatory support around non-traditional clinical study designs. According to a study done showed that Regulatory/Compliance Risk was cited as a major concern for decentralized clinical trials. Based on the events in the regulatory scene, the compliance and acceptance of the designs should be over in time.
Do not ignore traditional sites
With a flexible approach to clinical trials, sponsors look to conduct trials in both physical and remote sites. This means sponsors have to offer training, communication, and support in physical sites. Research shows that most investigators are unfamiliar with how remote trials work. So take advantage of the physical sites to trade the remote trial workers.
The clinical trial ecosystem is changing at a rapid pace. Before designing a decentralized clinical trial, be sure to consider all factors to ensure it works seamlessly.
Infographic provided by The Emmes Company, a clinical research services organization